"Control is good, trust is better – at least when it comes to personnel matters"
Hannah Zeichhardt, CEO

Because competent employees only work consistently at the highest level if they are happy. And the highest level is always our goal.
Send your application with as many relevant documents as possible to:
EmailUnsolicited applications are always welcome – whether for work in the laboratory, for planning and evaluation, for management and administration or for a mini-job.

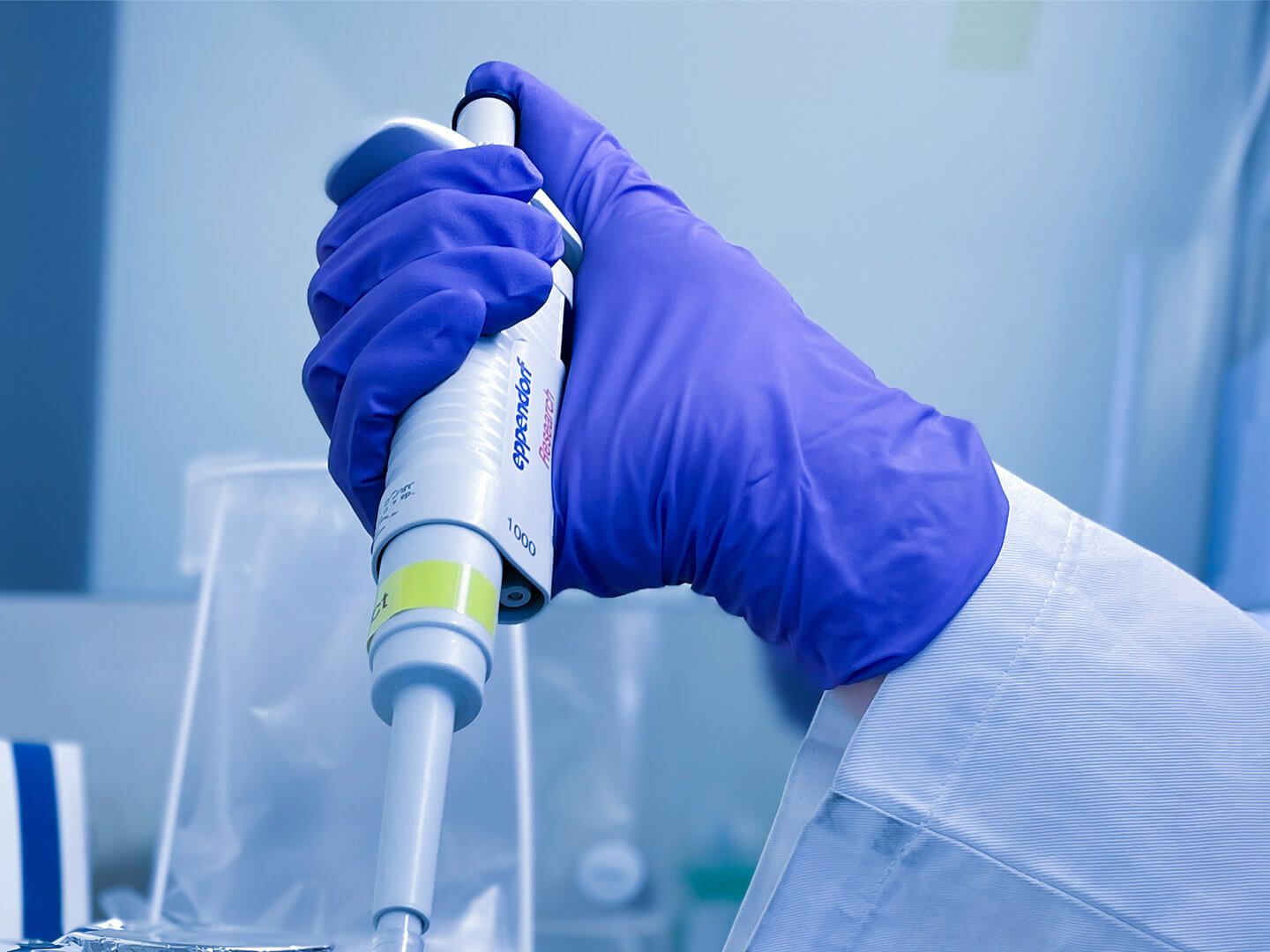
Our laboratory requires a wide range of expertise and specialist knowledge. And the motivation to think, produce and manage a sample from the idea to the last data record. There is hardly any monotony here! MTLA, MFA, biotechnologists and laboratory assistants work as a team for our common cause.

We produce quality control samples – for this we need people who can plan well, like to analyze correlations, juggle with data and want to automate processes. Together, we combine our strengths for our colleagues and customers.

We are only as good as our supporting processes allow us to be. That's why our organizational talents ensure that all processes run as smoothly as possible. Enjoyment of defined processes is just as important here as courage and friendliness towards everything new.
Our "hard-working helpers", who provide support in stressful phases or help with structure and organization, create space for change.